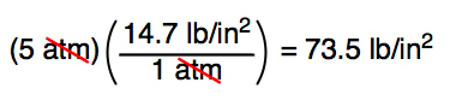SOLVED:Make the indicated pressure conversions. a. 17.3 psi to kilopascals b. 1.15 atm to psi c. 4.25 atm to mm Hg d. 224 psi to atmospheres

SOLVED:Make the indicated pressure conversions. a. 6.42 atm to kilopascals b. 4.21 atm to torr c. 794 mm Hg to atmospheres d. 27.2 psi to atmospheres

SOLVED: The atmosphere on Venus is much denser than on Earth: As a result;, Venusian atmospheric pressure is 1.35 x 103 psi. What is this pressure in atm?

Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download
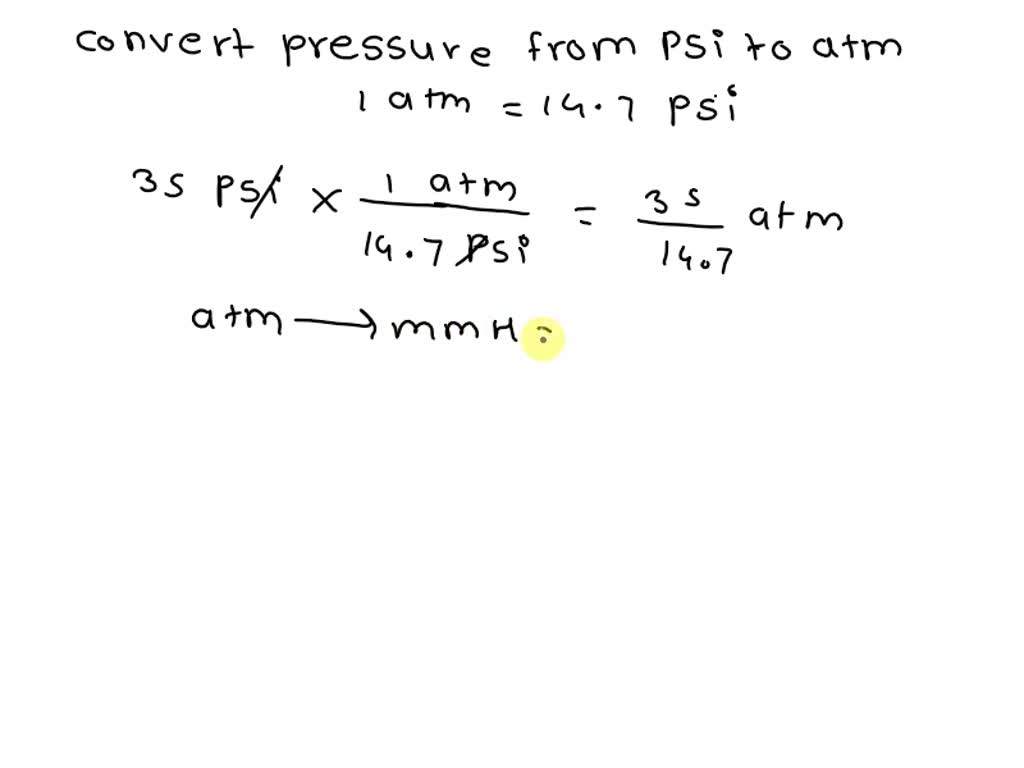
SOLVED: The recommended pressure for inflation of an automobile tire is 35 psi. What is the pressure in units of mm Hg? (1 atm=14.7 psi ; 1 atm=760 mmHg)(LO 10.1) (a) 3.9 ×